|
We expect six fundamental vibrations (12 minus 6), and these have been assigned to the spectrum absorptions.On the immediate high energy side of the visible spectrum lies the ultraviolet, and on the low energy side is the infrared.The portion of the infrared region most useful for analysis of organic compounds is not immediately adjacent to the visible spectrum, but is that having a wavelength range from 2,500 to 16,000 nm, with a corresponding frequency range from 1.910 13 to 1.210 14 Hz.The covalent bonds in molecules are not rigid sticks or rods, such as found in molecular model kits, but are more like stiff springs that can be stretched and bent.
The mobile nature of organic molecules was noted in the chapter concerning conformational isomers. We must now recognize that, in addition to the facile rotation of groups about single bonds, molecules experience a wide variety of vibrational motions, characteristic of their component atoms. 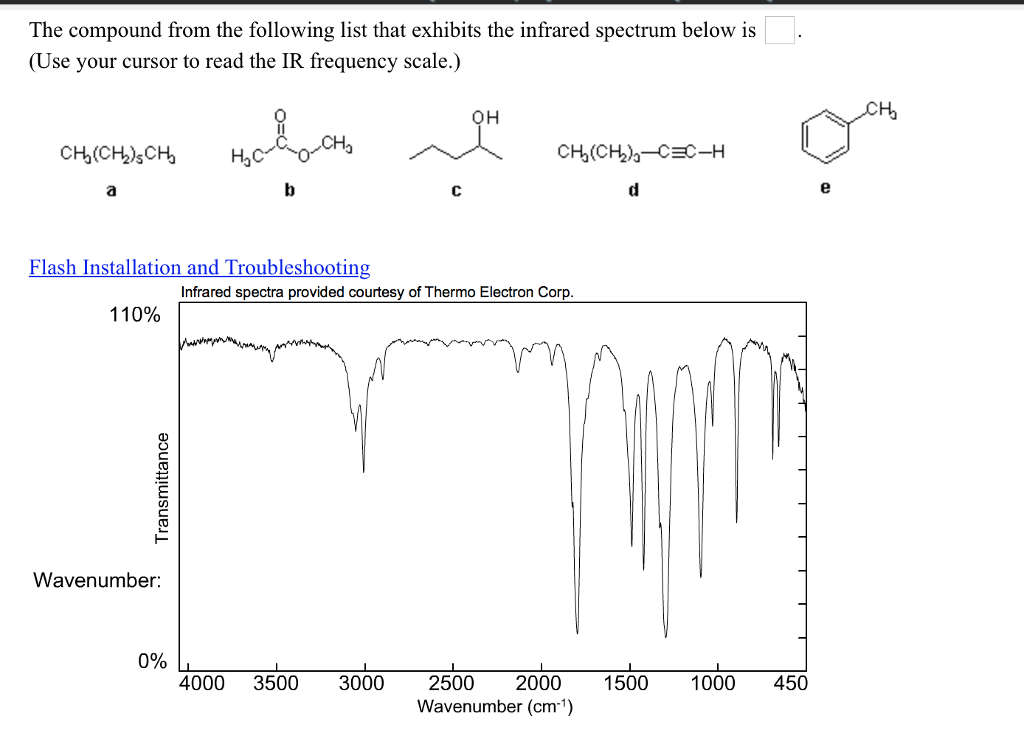
Infrared spectrometers, similar in principle to the UV-Visible spectrometer described elsewhere, permit chemists to obtain absorption spectra of compounds that are a unique reflection of their molecular structure. An example of such a spectrum is that of the flavoring agent vanillin, shown below. The gap in the spectrum between 700 800 cm -1 is due to solvent (CCl 4 ) absorption. Further analysis (below) will show that this spectrum also indicates the presence of an aldehyde function, a phenolic hydroxyl and a substituted benzene ring. The inverted display of absorption, compared with UV-Visible spectra, is characteristic. Thus a sample that did not absorb at all would record a horizontal line at 100 transmittance (top of the chart). The reciprocal centimeter is the number of wave cycles in one centimeter; whereas, frequency in cycles per second or Hz is equal to the number of wave cycles in 310 10 cm (the distance covered by light in one second). Wavelength units are in micrometers, microns (), instead of nanometers for the same reason. Most infrared spectra are displayed on a linear frequency scale, as shown here, but in some older texts a linear wavelength scale is used. A calculator for interconverting these frequency and wavelength values is provided on the right. Simply enter the value to be converted in the appropriate box, press Calculate and the equivalent number will appear in the empty box. Infrared spectra may be obtained from samples in all phases (liquid, solid and gaseous). Liquids are usually examined as a thin film sandwiched between two polished salt plates (note that glass absorbs infrared radiation, whereas NaCl is transparent). If solvents are used to dissolve solids, care must be taken to avoid obscuring important spectral regions by solvent absorption. Perchlorinated solvents such as carbon tetrachloride, chloroform and tetrachloroethene are commonly used. Alternatively, solids may either be incorporated in a thin KBr disk, prepared under high pressure, or mixed with a little non-volatile liquid and ground to a paste (or mull) that is smeared between salt plates. This leaves 3n-6 degrees of vibrational freedom (3n-5 if the molecule is linear). Vibrational modes are often given descriptive names, such as stretching, bending, scissoring, rocking and twisting. The four-atom molecule of formaldehyde, the gas phase spectrum of which is shown below, provides an example of these terms. If a ball stick model of formaldehyde is not displayed to the right of the spectrum, press the view ballstick model button on the right.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Bartender 10-1 product key code sr4
- Blog
- 2011 expedition navigation map update
- Colin mcrae rally 2005
- Michael jackson man in the mirror official video
- Ask and it is given listed under
- Kurtis blow basketball tv show
- Best root stimulator for trees
- Veeam backup restore
- Mii channel theme launchpad
- Helpconsole darkroom booth user guide default
- Una paloma blanca song lyrics
- How to change aula mouse backlit light colors
- Reimage licence key serial

 RSS Feed
RSS Feed
